Call it a kick up the backside.
For decades, Europe has failed to address its biopharma weaknesses – too little lab-to-drug translation, too little scale-up capital, non-existent public markets and penny-pinching drug budgets.
Then came President Trump. Love him or hate him, his call-out of the US-EU drug pricing gap, his tariff-talk and his efforts to onshore US manufacturing have woken Europe up. “Heads of state are starting to realize there’s a new reality,” said Ipsen CEO David Loew during the keynote session at Bio-Europe in Vienna on November 3.
Dealing with that new reality means stepping up – fast. “Europe has an incredible base of intellectual power and fundamental biopharma research,” said Jeremy Levin, chairman emeritus at US advocacy group BIO and CEO of Ovid Therapeutics. “But you’ve been dependent on Big Brother.”
To de-couple, Europe must pay more for newly launched drugs, learn from China’s superior R&D efficiency (rather than worry about it), re-design reward structures for academic researchers and compel its pension funds to invest more into innovation at home. Lastly, European citizens need to understand more clearly the value of a strong biopharma sector – in improving health, saving lives and growing economies.
“We need to communicate more directly with the European people,” said Loew. “Biotech voices have not been heard loudly enough.” Dismal pharma industry PR and a few bad actors have left the sector better known for price-gouging than for generating jobs, factories, labs, wealth and health.
These messages aren’t new. But they are now being heard by Europe’s policymakers and will soon be bellowed out to the people: EFPIA is apparently planning an “aggressive” outreach campaign.
1. Higher drug prices
The most prominent of Trump’s drug policy antics is Most-Favored-Nation (MFN) pricing, which ties US Medicaid drug launch prices to those in a handful of other developed economies including the big five EU countries, Denmark, Switzerland and Japan. As Big Pharma CEOs queue up to sign MFN deals with a US president intent on reducing US-EU price disparity, Europe’s national leaders “understand they will probably have to raise prices for newly launched drugs,” said Loew. Meetings are happening, he added, with Germany’s Chancellor Merz, French president Macron, UK prime minister Keir Starmer and the European Commission’s Health and Food Safety Commissioner Oliver Varhelyi.
Biotech leaders may have to change their business development playbook. MFN deals will make it harder for biotechs to out-license ex-US right to their drugs without keeping a tight leash on partner pricing strategies. Future territory deals will likely include new contractual clauses restricting price flexibility and/or more joined-up thinking among partners; consultants will make hay advising on launch sequence strategies.
For now, though, MFN deals involve only Medicaid (the US state program for low-income Americans) and apply only to new drug launches. Just a handful of deals have been signed (including Pfizer, AstraZeneca and Merck). “It’s going to take years to significantly raise European drug prices,” said Loew.
“So you have time.”
2. Learn from China
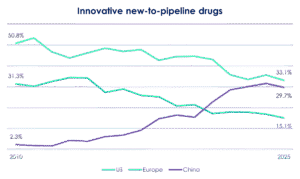
Where Europe doesn’t have time, though, is in figuring out how to compete with China. The writing has long been on the wall, Levin told the Bio-Europe audience. “Twenty-five years ago, China said it would do [grow its life sciences sector], and it has re-iterated that every five years since,” he said. The result: China’s share of pipeline assets in 2025 has overtaken Europe’s, according to Evaluate data presented by Daniel Chancellor. The country’s share of licensing deal value, less than 5% in 2020, has multiplied to almost 40%. “This growth will continue to accelerate,” warned Levin.
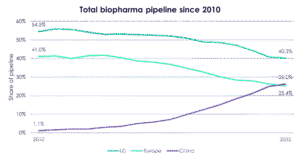
Source: Evaluate Pharma
But China will only dominate if Europe lets it, Levin continued. China is good at rapidly engineering more, sometimes better, drug candidates out of Europe and US discoveries. But it’s less good – for now – at brand new research.
3. Incentivize lab-to-drug translation
That will change. In the meantime, Europe should look to China to become more efficient, faster, cheaper and better itself, said Loew. Europe must create a more drug-development-friendly environment that more directly encourages brilliant research to be translated at home into brilliant drugs. “University professors are often rewarded based on publications. But if those are picked up by others who build biotechs in the US or China, we haven’t really done our job” in Europe, said Loew.
Academics’ reward structures should be re-designed, he said, such that more funding for professors and post-docs is tied to a lab or research group’s royalty income, rather than state hand-outs.
4. Tap pension funds
Funding translation requires more scale-up capital. Tapping into European pension funds to invest more at home is a well-aired idea to address that shortfall, but action has been slow.
Now, with the White House having “shaken the [European] tree,” things may accelerate, said Loew. Individual countries including Britain are already trying to shift pension legislation. They might also take a leaf out of Australia’s book. Levin pointed to that country’s national pension fund, one of the largest on a per-capita basis. It invests tens of billions of dollars into innovative Australian companies, as part of a local investment strategy. The result? An explosion of domestic innovation in the last four or five years.
Raising more capital in Europe must be accompanied by less red tape. Biopharma is identified as ‘strategic’ in many national growth strategy documents. But sticky employment and immigration laws, heavy clinical trial bureaucracy and burdensome planning and building rules in many countries continue to hold it back.
Happily, at the European level, the Commission is starting to back pedal on “completely crazy” new pharmaceutical legislation that would have shortened data protection periods and constrained drug launch flexibility across the continent, said Loew. The European Commission’s Health Directorate “is more open now that any time over the last twenty years,” he said. Commissioner Varhelyi is “acutely aware that things need to change.”
5. Tell the people [how much Switzerland pays for its cows]
Advocacy must happen regionally, nationally – and at the individual level. “It’s time to wake up Europe’s population to the wealth-generating power of biopharma,” said Loew.
The panelists called for louder engagement with the people in determining national budget allocation. “Educated people will make the right decisions,” said Loew.
Hopefully. Yet perhaps the most memorable figure from the 90 minute conversation was how much Switzerland used to invest per cow, relative to its investment in citizens’ health.
“Switzerland spent about €4000 per cow, per year,” said Loew, recalling his time at Roche during the 2000s. “The equivalent figure per [human] head, on cancer drugs, was €80.”
That was in 2007. Nearly twenty years later, amid what Levin calls Trump’s “complete revamp of the American social system,” this US president may just have catalyzed European biopharma into action.
